The second ionization energy is defined as the energy needed to remove an electron from one mole of 1+ ions in the gaseous state to form one mole of 2+ ions in the gaseous state.
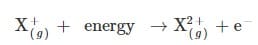
Down the group, the ionization energies decrease with increase in atomic radii.
Order of ionization energies in IA group ——-> Li > Na > K > Rb > Cs > Fr
Ionization energy increases with increase in the effective nuclear charge.
Ionization energy decreases with increase in screening power of inner electrons.
The order of screening power of electrons present in different types of orbitals is: s > p > d > f .
Greater the penetration power, greater will be the ionization energy.
The order of penetration power of electrons in different orbitals is as follows.
s > p > d > f
Atoms with stable electronic configurations like: ns2 np6 or half filled or completely filled sub shells are extra stable. Removal of electrons from them is difficult and hence their ionization energies are comparatively high.
These configurations are more symmetric and the exchange energy is minimal. Hence they are relatively more stable.
E.g. Noble gases with ns2 np6 ; IIA group elements with ns2 (completely filled); and VA group elements with ns2 np3 (half filled) configurations have higher ionization potentials.
